GO Annotation
| Category | Term ID | Term description |
|---|---|---|
| Process | GO:0000122 | Negative regulation of transcription by RNA polymerase II |
| Process | GO:0001568 | Blood vessel development |
| Process | GO:0001659 | Temperature homeostasis |
| Process | GO:0001678 | Cellular glucose homeostasis |
| Process | GO:0001944 | Vasculature development |
| Process | GO:0002791 | Regulation of peptide secretion |
| Process | GO:0002792 | Negative regulation of peptide secretion |
| Process | GO:0006109 | Regulation of carbohydrate metabolic process |
| Process | GO:0006111 | Regulation of gluconeogenesis |
| Process | GO:0006355 | Regulation of transcription, DNA-templated |
| Process | GO:0006357 | Regulation of transcription by RNA polymerase II |
| Process | GO:0006473 | Protein acetylation |
| Process | GO:0006807 | Nitrogen compound metabolic process |
| Process | GO:0006914 | Autophagy |
| Process | GO:0006915 | Apoptotic process |
| Process | GO:0006950 | Response to stress |
| Process | GO:0006974 | Cellular response to DNA damage stimulus |
| Process | GO:0006979 | Response to oxidative stress |
| Process | GO:0007154 | Cell communication |
| Process | GO:0007165 | Signal transduction |
| Process | GO:0007166 | Cell surface receptor signaling pathway |
| Process | GO:0007167 | Enzyme-linked receptor protein signaling pathway |
| Process | GO:0007169 | Transmembrane receptor protein tyrosine kinase signaling pathway |
| Process | GO:0007267 | Cell-cell signaling |
| Process | GO:0007275 | Multicellular organism development |
| Process | GO:0008152 | Metabolic process |
| Process | GO:0008219 | Cell death |
| Process | GO:0008286 | Insulin receptor signaling pathway |
| Process | GO:0009056 | Catabolic process |
| Process | GO:0009266 | Response to temperature stimulus |
| Process | GO:0009267 | Cellular response to starvation |
| Process | GO:0009409 | Response to cold |
| Process | GO:0009605 | Response to external stimulus |
| Process | GO:0009628 | Response to abiotic stimulus |
| Process | GO:0009719 | Response to endogenous stimulus |
| Process | GO:0009725 | Response to hormone |
| Process | GO:0009889 | Regulation of biosynthetic process |
| Process | GO:0009890 | Negative regulation of biosynthetic process |
| Process | GO:0009891 | Positive regulation of biosynthetic process |
| Process | GO:0009892 | Negative regulation of metabolic process |
| Process | GO:0009893 | Positive regulation of metabolic process |
| Process | GO:0009894 | Regulation of catabolic process |
| Process | GO:0009896 | Positive regulation of catabolic process |
| Process | GO:0009966 | Regulation of signal transduction |
| Process | GO:0009968 | Negative regulation of signal transduction |
| Process | GO:0009987 | Cellular process |
| Process | GO:0009991 | Response to extracellular stimulus |
| Process | GO:0010033 | Response to organic substance |
| Process | GO:0010035 | Response to inorganic substance |
| Process | GO:0010243 | Response to organonitrogen compound |
| Process | GO:0010468 | Regulation of gene expression |
| Process | GO:0010506 | Regulation of autophagy |
| Process | GO:0010508 | Positive regulation of autophagy |
| Process | GO:0010556 | Regulation of macromolecule biosynthetic process |
| Process | GO:0010557 | Positive regulation of macromolecule biosynthetic process |
| Process | GO:0010558 | Negative regulation of macromolecule biosynthetic process |
| Process | GO:0010604 | Positive regulation of macromolecule metabolic process |
| Process | GO:0010605 | Negative regulation of macromolecule metabolic process |
| Process | GO:0010611 | Regulation of cardiac muscle hypertrophy |
| Process | GO:0010612 | Regulation of cardiac muscle adaptation |
| Process | GO:0010614 | Negative regulation of cardiac muscle hypertrophy |
| Process | GO:0010616 | Negative regulation of cardiac muscle adaptation |
| Process | GO:0010646 | Regulation of cell communication |
| Process | GO:0010648 | Negative regulation of cell communication |
| Process | GO:0010675 | Regulation of cellular carbohydrate metabolic process |
| Process | GO:0010676 | Positive regulation of cellular carbohydrate metabolic process |
| Process | GO:0010817 | Regulation of hormone levels |
| Process | GO:0010906 | Regulation of glucose metabolic process |
| Process | GO:0010907 | Positive regulation of glucose metabolic process |
| Process | GO:0010941 | Regulation of cell death |
| Process | GO:0010942 | Positive regulation of cell death |
| Process | GO:0012501 | Programmed cell death |
| Process | GO:0014741 | Negative regulation of muscle hypertrophy |
| Process | GO:0014743 | Regulation of muscle hypertrophy |
| Process | GO:0014745 | Negative regulation of muscle adaptation |
| Process | GO:0016055 | Wnt signaling pathway |
| Process | GO:0019219 | Regulation of nucleobase-containing compound metabolic process |
| Process | GO:0019222 | Regulation of metabolic process |
| Process | GO:0019538 | Protein metabolic process |
| Process | GO:0019725 | Cellular homeostasis |
| Process | GO:0019827 | Stem cell population maintenance |
| Process | GO:0023051 | Regulation of signaling |
| Process | GO:0023052 | Signaling |
| Process | GO:0023057 | Negative regulation of signaling |
| Process | GO:0030111 | Regulation of Wnt signaling pathway |
| Process | GO:0030154 | Cell differentiation |
| Process | GO:0030178 | Negative regulation of Wnt signaling pathway |
| Process | GO:0031323 | Regulation of cellular metabolic process |
| Process | GO:0031324 | Negative regulation of cellular metabolic process |
| Process | GO:0031325 | Positive regulation of cellular metabolic process |
| Process | GO:0031326 | Regulation of cellular biosynthetic process |
| Process | GO:0031327 | Negative regulation of cellular biosynthetic process |
| Process | GO:0031328 | Positive regulation of cellular biosynthetic process |
| Process | GO:0031329 | Regulation of cellular catabolic process |
| Process | GO:0031331 | Positive regulation of cellular catabolic process |
| Process | GO:0031667 | Response to nutrient levels |
| Process | GO:0031668 | Cellular response to extracellular stimulus |
| Process | GO:0031669 | Cellular response to nutrient levels |
| Process | GO:0032501 | Multicellular organismal process |
| Process | GO:0032502 | Developmental process |
| Process | GO:0032868 | Response to insulin |
| Process | GO:0032869 | Cellular response to insulin stimulus |
| Process | GO:0032870 | Cellular response to hormone stimulus |
| Process | GO:0032872 | Regulation of stress-activated MAPK cascade |
| Process | GO:0032873 | Negative regulation of stress-activated MAPK cascade |
| Process | GO:0032879 | Regulation of localization |
| Process | GO:0032880 | Regulation of protein localization |
| Process | GO:0033500 | Carbohydrate homeostasis |
| Process | GO:0033554 | Cellular response to stress |
| Process | GO:0033993 | Response to lipid |
| Process | GO:0034599 | Cellular response to oxidative stress |
| Process | GO:0036211 | Protein modification process |
| Process | GO:0036295 | Cellular response to increased oxygen levels |
| Process | GO:0036296 | Response to increased oxygen levels |
| Process | GO:0042127 | Regulation of cell population proliferation |
| Process | GO:0042176 | Regulation of protein catabolic process |
| Process | GO:0042221 | Response to chemical |
| Process | GO:0042592 | Homeostatic process |
| Process | GO:0042593 | Glucose homeostasis |
| Process | GO:0042594 | Response to starvation |
| Process | GO:0042981 | Regulation of apoptotic process |
| Process | GO:0043065 | Positive regulation of apoptotic process |
| Process | GO:0043066 | Negative regulation of apoptotic process |
| Process | GO:0043067 | Regulation of programmed cell death |
| Process | GO:0043068 | Positive regulation of programmed cell death |
| Process | GO:0043069 | Negative regulation of programmed cell death |
| Process | GO:0043170 | Macromolecule metabolic process |
| Process | GO:0043255 | Regulation of carbohydrate biosynthetic process |
| Process | GO:0043408 | Regulation of MAPK cascade |
| Process | GO:0043409 | Negative regulation of MAPK cascade |
| Process | GO:0043412 | Macromolecule modification |
| Process | GO:0043434 | Response to peptide hormone |
| Process | GO:0043502 | Regulation of muscle adaptation |
| Process | GO:0043543 | Protein acylation |
| Process | GO:0044057 | Regulation of system process |
| Process | GO:0044237 | Cellular metabolic process |
| Process | GO:0044238 | Primary metabolic process |
| Process | GO:0044248 | Cellular catabolic process |
| Process | GO:0045444 | Fat cell differentiation |
| Process | GO:0045595 | Regulation of cell differentiation |
| Process | GO:0045596 | Negative regulation of cell differentiation |
| Process | GO:0045598 | Regulation of fat cell differentiation |
| Process | GO:0045599 | Negative regulation of fat cell differentiation |
| Process | GO:0045722 | Positive regulation of gluconeogenesis |
| Process | GO:0045732 | Positive regulation of protein catabolic process |
| Process | GO:0045892 | Negative regulation of transcription, DNA-templated |
| Process | GO:0045893 | Positive regulation of transcription, DNA-templated |
| Process | GO:0045913 | Positive regulation of carbohydrate metabolic process |
| Process | GO:0045934 | Negative regulation of nucleobase-containing compound metabolic process |
| Process | GO:0045935 | Positive regulation of nucleobase-containing compound metabolic process |
| Process | GO:0045944 | Positive regulation of transcription by RNA polymerase II |
| Process | GO:0046676 | Negative regulation of insulin secretion |
| Process | GO:0046883 | Regulation of hormone secretion |
| Process | GO:0046888 | Negative regulation of hormone secretion |
| Process | GO:0048518 | Positive regulation of biological process |
| Process | GO:0048519 | Negative regulation of biological process |
| Process | GO:0048522 | Positive regulation of cellular process |
| Process | GO:0048523 | Negative regulation of cellular process |
| Process | GO:0048583 | Regulation of response to stimulus |
| Process | GO:0048585 | Negative regulation of response to stimulus |
| Process | GO:0048731 | System development |
| Process | GO:0048856 | Anatomical structure development |
| Process | GO:0048869 | Cellular developmental process |
| Process | GO:0048871 | Multicellular organismal homeostasis |
| Process | GO:0048878 | Chemical homeostasis |
| Process | GO:0050708 | Regulation of protein secretion |
| Process | GO:0050709 | Negative regulation of protein secretion |
| Process | GO:0050789 | Regulation of biological process |
| Process | GO:0050793 | Regulation of developmental process |
| Process | GO:0050794 | Regulation of cellular process |
| Process | GO:0050796 | Regulation of insulin secretion |
| Process | GO:0050896 | Response to stimulus |
| Process | GO:0051046 | Regulation of secretion |
| Process | GO:0051048 | Negative regulation of secretion |
| Process | GO:0051049 | Regulation of transport |
| Process | GO:0051051 | Negative regulation of transport |
| Process | GO:0051093 | Negative regulation of developmental process |
| Process | GO:0051171 | Regulation of nitrogen compound metabolic process |
| Process | GO:0051172 | Negative regulation of nitrogen compound metabolic process |
| Process | GO:0051173 | Positive regulation of nitrogen compound metabolic process |
| Process | GO:0051223 | Regulation of protein transport |
| Process | GO:0051224 | Negative regulation of protein transport |
| Process | GO:0051239 | Regulation of multicellular organismal process |
| Process | GO:0051241 | Negative regulation of multicellular organismal process |
| Process | GO:0051246 | Regulation of protein metabolic process |
| Process | GO:0051247 | Positive regulation of protein metabolic process |
| Process | GO:0051252 | Regulation of RNA metabolic process |
| Process | GO:0051253 | Negative regulation of RNA metabolic process |
| Process | GO:0051254 | Positive regulation of RNA metabolic process |
| Process | GO:0051716 | Cellular response to stimulus |
| Process | GO:0055082 | Cellular chemical homeostasis |
| Process | GO:0055093 | Response to hyperoxia |
| Process | GO:0060070 | Canonical Wnt signaling pathway |
| Process | GO:0060255 | Regulation of macromolecule metabolic process |
| Process | GO:0060341 | Regulation of cellular localization |
| Process | GO:0060548 | Negative regulation of cell death |
| Process | GO:0060828 | Regulation of canonical Wnt signaling pathway |
| Process | GO:0061919 | Process utilizing autophagic mechanism |
| Process | GO:0062012 | Regulation of small molecule metabolic process |
| Process | GO:0062013 | Positive regulation of small molecule metabolic process |
| Process | GO:0062197 | Cellular response to chemical stress |
| Process | GO:0065007 | Biological regulation |
| Process | GO:0065008 | Regulation of biological quality |
| Process | GO:0070201 | Regulation of establishment of protein localization |
| Process | GO:0070302 | Regulation of stress-activated protein kinase signaling cascade |
| Process | GO:0070303 | Negative regulation of stress-activated protein kinase signaling cascade |
| Process | GO:0070417 | Cellular response to cold |
| Process | GO:0070482 | Response to oxygen levels |
| Process | GO:0070542 | Response to fatty acid |
| Process | GO:0070887 | Cellular response to chemical stimulus |
| Process | GO:0071241 | Cellular response to inorganic substance |
| Process | GO:0071310 | Cellular response to organic substance |
| Process | GO:0071375 | Cellular response to peptide hormone stimulus |
| Process | GO:0071417 | Cellular response to organonitrogen compound |
| Process | GO:0071453 | Cellular response to oxygen levels |
| Process | GO:0071455 | Cellular response to hyperoxia |
| Process | GO:0071495 | Cellular response to endogenous stimulus |
| Process | GO:0071496 | Cellular response to external stimulus |
| Process | GO:0071704 | Organic substance metabolic process |
| Process | GO:0071731 | Response to nitric oxide |
| Process | GO:0071732 | Cellular response to nitric oxide |
| Process | GO:0072359 | Circulatory system development |
| Process | GO:0080090 | Regulation of primary metabolic process |
| Process | GO:0080134 | Regulation of response to stress |
| Process | GO:0080135 | Regulation of cellular response to stress |
| Process | GO:0090087 | Regulation of peptide transport |
| Process | GO:0090090 | Negative regulation of canonical Wnt signaling pathway |
| Process | GO:0090257 | Regulation of muscle system process |
| Process | GO:0090276 | Regulation of peptide hormone secretion |
| Process | GO:0090278 | Negative regulation of peptide hormone secretion |
| Process | GO:0097009 | Energy homeostasis |
| Process | GO:0097150 | Neuronal stem cell population maintenance |
| Process | GO:0098727 | Maintenance of cell number |
| Process | GO:0198738 | Cell-cell signaling by wnt |
| Process | GO:1901564 | Organonitrogen compound metabolic process |
| Process | GO:1901652 | Response to peptide |
| Process | GO:1901653 | Cellular response to peptide |
| Process | GO:1901698 | Response to nitrogen compound |
| Process | GO:1901699 | Cellular response to nitrogen compound |
| Process | GO:1901700 | Response to oxygen-containing compound |
| Process | GO:1901701 | Cellular response to oxygen-containing compound |
| Process | GO:1902170 | Cellular response to reactive nitrogen species |
| Process | GO:1902531 | Regulation of intracellular signal transduction |
| Process | GO:1902532 | Negative regulation of intracellular signal transduction |
| Process | GO:1902679 | Negative regulation of RNA biosynthetic process |
| Process | GO:1902680 | Positive regulation of RNA biosynthetic process |
| Process | GO:1903242 | Regulation of cardiac muscle hypertrophy in response to stress |
| Process | GO:1903243 | Negative regulation of cardiac muscle hypertrophy in response to stress |
| Process | GO:1903506 | Regulation of nucleic acid-templated transcription |
| Process | GO:1903507 | Negative regulation of nucleic acid-templated transcription |
| Process | GO:1903508 | Positive regulation of nucleic acid-templated transcription |
| Process | GO:1903530 | Regulation of secretion by cell |
| Process | GO:1903531 | Negative regulation of secretion by cell |
| Process | GO:1903828 | Negative regulation of protein localization |
| Process | GO:1904950 | Negative regulation of establishment of protein localization |
| Process | GO:1905114 | Cell surface receptor signaling pathway involved in cell-cell signaling |
| Process | GO:2000177 | Regulation of neural precursor cell proliferation |
| Process | GO:2000377 | Regulation of reactive oxygen species metabolic process |
| Process | GO:2001141 | Regulation of RNA biosynthetic process |
| Component | GO:0000785 | Chromatin |
| Component | GO:0005622 | Intracellular anatomical structure |
| Component | GO:0005634 | Nucleus |
| Component | GO:0005654 | Nucleoplasm |
| Component | GO:0005694 | Chromosome |
| Component | GO:0005737 | Cytoplasm |
| Component | GO:0005739 | Mitochondrion |
| Component | GO:0005829 | Cytosol |
| Component | GO:0031974 | Membrane-enclosed lumen |
| Component | GO:0031981 | Nuclear lumen |
| Component | GO:0043226 | Organelle |
| Component | GO:0043227 | Membrane-bounded organelle |
| Component | GO:0043228 | Non-membrane-bounded organelle |
| Component | GO:0043229 | Intracellular organelle |
| Component | GO:0043231 | Intracellular membrane-bounded organelle |
| Component | GO:0043232 | Intracellular non-membrane-bounded organelle |
| Component | GO:0043233 | Organelle lumen |
| Component | GO:0070013 | Intracellular organelle lumen |
| Component | GO:0110165 | Cellular anatomical entity |
| Function | GO:0000976 | Transcription cis-regulatory region binding |
| Function | GO:0000977 | RNA polymerase II transcription regulatory region sequence-specific DNA binding |
| Function | GO:0000978 | RNA polymerase II cis-regulatory region sequence-specific DNA binding |
| Function | GO:0000981 | DNA-binding transcription factor activity, RNA polymerase II-specific |
| Function | GO:0000987 | Cis-regulatory region sequence-specific DNA binding |
| Function | GO:0001067 | Transcription regulatory region nucleic acid binding |
| Function | GO:0001216 | DNA-binding transcription activator activity |
| Function | GO:0001217 | DNA-binding transcription repressor activity |
| Function | GO:0001227 | DNA-binding transcription repressor activity, RNA polymerase II-specific |
| Function | GO:0001228 | DNA-binding transcription activator activity, RNA polymerase II-specific |
| Function | GO:0003676 | Nucleic acid binding |
| Function | GO:0003677 | DNA binding |
| Function | GO:0003682 | Chromatin binding |
| Function | GO:0003690 | Double-stranded DNA binding |
| Function | GO:0003700 | DNA-binding transcription factor activity |
| Function | GO:0005488 | Binding |
| Function | GO:0005515 | Protein binding |
| Function | GO:0008013 | Beta-catenin binding |
| Function | GO:0019899 | Enzyme binding |
| Function | GO:0019902 | Phosphatase binding |
| Function | GO:0019903 | Protein phosphatase binding |
| Function | GO:0031490 | Chromatin DNA binding |
| Function | GO:0031625 | Ubiquitin protein ligase binding |
| Function | GO:0043565 | Sequence-specific DNA binding |
| Function | GO:0044389 | Ubiquitin-like protein ligase binding |
| Function | GO:0051721 | Protein phosphatase 2A binding |
| Function | GO:0097159 | Organic cyclic compound binding |
| Function | GO:0140110 | Transcription regulator activity |
| Function | GO:1901363 | Heterocyclic compound binding |
| Function | GO:1990837 | Sequence-specific double-stranded DNA binding |
| Function | GO:1990841 | Promoter-specific chromatin binding |
Pathway annotation
Expression profile of FOXO1 in omics data
| ID | GSE101724 |
| Title | Effects of Propofol Treatment in Neural Progenitors Derived from Human Induced Pluripotent Stem Cells |
| Organism | Homo sapiens |
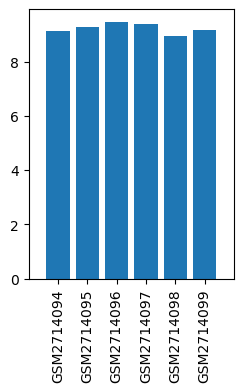
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM2714094 | untreated rep1 | treatment: untreated | 9.117643309 |
| GSM2714095 | untreated rep2 | treatment: untreated | 9.290491252 |
| GSM2714096 | 20 uM propofol rep1 | treatment: 20 uM propofol | 9.459573342 |
| GSM2714097 | 20 uM propofol rep2 | treatment: 20 uM propofol | 9.38274876 |
| GSM2714098 | 300 uM propofol rep1 | treatment: 300 uM propofol | 8.937681185 |
| GSM2714099 | 300 uM propofol rep2 | treatment: 300 uM propofol | 9.146704733 |
| ID | GSE120596 |
| Title | Variability in the Analgesic Response to Ibuprofen Following Third Molar Extraction is Associated with Differences in Activation of the Cyclooxygenase Pathway |
| Organism | Homo sapiens |
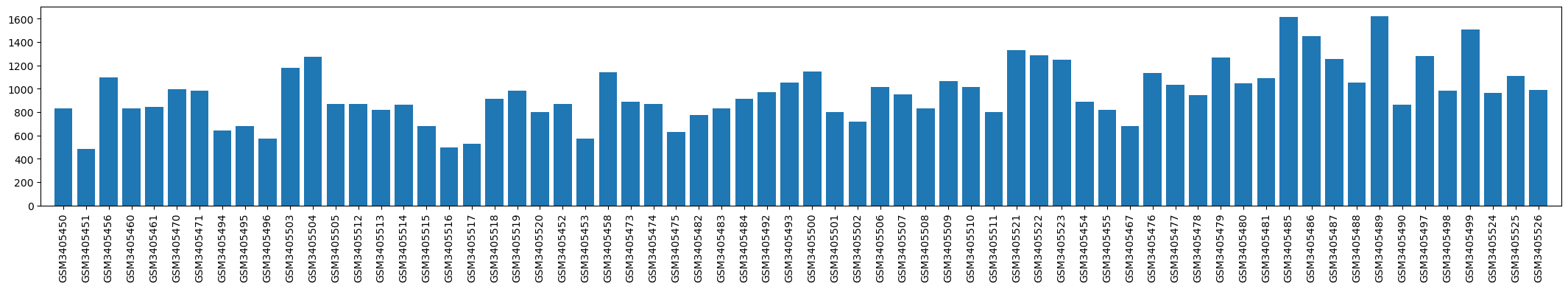
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM3405450 | 1001_baseline | cell type: peripheral blood mononuclear cell subject: 1001 gender: Female timepoint: baseline library barcode: ATTACTCG-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | 833.0 |
| GSM3405451 | 1001_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1001 gender: Female timepoint: post-surgery 1 library barcode: TCCGGAGA-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | 487.0 |
| GSM3405452 | 1002_baseline | cell type: peripheral blood mononuclear cell subject: 1002 gender: Female timepoint: baseline library barcode: CGCTCATT-TATAGCCT drug treatment: Placebo response group: Placebo | 867.0 |
| GSM3405453 | 1002_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1002 gender: Female timepoint: post-surgery 1 library barcode: GAGATTCC-TATAGCCT drug treatment: Placebo response group: Placebo | 570.0 |
| GSM3405454 | 1003_baseline | cell type: peripheral blood mononuclear cell subject: 1003 gender: Male timepoint: baseline library barcode: ATTCAGAA-TATAGCCT drug treatment: ibuprofen sodium response group: Full responder | 890.0 |
| GSM3405455 | 1003_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1003 gender: Male timepoint: post-surgery 1 library barcode: GAATTCGT-TATAGCCT drug treatment: ibuprofen sodium response group: Full responder | 821.0 |
| GSM3405456 | 1004_baseline | cell type: peripheral blood mononuclear cell subject: 1004 gender: Male timepoint: baseline library barcode: CTGAAGCT-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | 1094.0 |
| GSM3405457 | 1004_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1004 gender: Male timepoint: post-surgery 1 library barcode: TAATGCGC-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | |
| GSM3405458 | 1005_baseline | cell type: peripheral blood mononuclear cell subject: 1005 gender: Male timepoint: baseline library barcode: CGGCTATG-TATAGCCT drug treatment: Placebo response group: Placebo | 1139.0 |
| GSM3405459 | 1005_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1005 gender: Male timepoint: post-surgery 1 library barcode: TCCGCGAA-TATAGCCT drug treatment: Placebo response group: Placebo | |
| GSM3405460 | 1006_baseline | cell type: peripheral blood mononuclear cell subject: 1006 gender: Male timepoint: baseline library barcode: TCTCGCGC-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | 831.0 |
| GSM3405461 | 1006_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1006 gender: Male timepoint: post-surgery 1 library barcode: AGCGATAG-TATAGCCT drug treatment: ibuprofen sodium response group: Partial responder | 846.0 |
| GSM3405462 | 1007_baseline | cell type: peripheral blood mononuclear cell subject: 1007 gender: Male timepoint: baseline library barcode: ATTACTCG-ATAGAGGC drug treatment: ibuprofen sodium response group: Full responder | |
| GSM3405463 | 1007_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1007 gender: Male timepoint: post-surgery 1 library barcode: TCCGGAGA-ATAGAGGC drug treatment: ibuprofen sodium response group: Full responder | |
| GSM3405464 | 1008_baseline | cell type: peripheral blood mononuclear cell subject: 1008 gender: Male timepoint: baseline library barcode: CGCTCATT-ATAGAGGC drug treatment: Placebo response group: Placebo | |
| GSM3405465 | 1008_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1008 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-ATAGAGGC drug treatment: Placebo response group: Placebo | |
| GSM3405466 | 1008_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1008 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-ATAGAGGC drug treatment: Placebo response group: Placebo | |
| GSM3405467 | 1011_baseline | cell type: peripheral blood mononuclear cell subject: 1011 gender: Female timepoint: baseline library barcode: GAATTCGT-ATAGAGGC drug treatment: ibuprofen sodium response group: Full responder | 680.0 |
| GSM3405468 | 1011_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1011 gender: Female timepoint: post-surgery 1 library barcode: CTGAAGCT-ATAGAGGC drug treatment: ibuprofen sodium response group: Full responder | |
| GSM3405469 | 1011_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1011 gender: Female timepoint: post-surgery 2 library barcode: TAATGCGC-ATAGAGGC drug treatment: ibuprofen sodium response group: Full responder | |
| GSM3405470 | 1012_baseline | cell type: peripheral blood mononuclear cell subject: 1012 gender: Female timepoint: baseline library barcode: CGGCTATG-ATAGAGGC drug treatment: ibuprofen sodium response group: Partial responder | 993.0 |
| GSM3405471 | 1012_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1012 gender: Female timepoint: post-surgery 1 library barcode: TCCGCGAA-ATAGAGGC drug treatment: ibuprofen sodium response group: Partial responder | 981.0 |
| GSM3405472 | 1012_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1012 gender: Female timepoint: post-surgery 2 library barcode: TCTCGCGC-ATAGAGGC drug treatment: ibuprofen sodium response group: Partial responder | |
| GSM3405473 | 1014_baseline | cell type: peripheral blood mononuclear cell subject: 1014 gender: Male timepoint: baseline library barcode: AGCGATAG-ATAGAGGC drug treatment: Placebo response group: Placebo | 887.0 |
| GSM3405474 | 1014_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1014 gender: Male timepoint: post-surgery 1 library barcode: ATTACTCG-CCTATCCT drug treatment: Placebo response group: Placebo | 868.0 |
| GSM3405475 | 1014_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1014 gender: Male timepoint: post-surgery 2 library barcode: TCCGGAGA-CCTATCCT drug treatment: Placebo response group: Placebo | 631.0 |
| GSM3405476 | 1015_baseline | cell type: peripheral blood mononuclear cell subject: 1015 gender: Male timepoint: baseline library barcode: CGCTCATT-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1132.0 |
| GSM3405477 | 1015_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1015 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1036.0 |
| GSM3405478 | 1015_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1015 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 945.0 |
| GSM3405479 | 1016_baseline | cell type: peripheral blood mononuclear cell subject: 1016 gender: Male timepoint: baseline library barcode: GAATTCGT-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1265.0 |
| GSM3405480 | 1016_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1016 gender: Male timepoint: post-surgery 1 library barcode: CTGAAGCT-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1045.0 |
| GSM3405481 | 1016_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1016 gender: Male timepoint: post-surgery 2 library barcode: TAATGCGC-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1088.0 |
| GSM3405482 | 1017_baseline | cell type: peripheral blood mononuclear cell subject: 1017 gender: Male timepoint: baseline library barcode: CGGCTATG-CCTATCCT drug treatment: Placebo response group: Placebo | 773.0 |
| GSM3405483 | 1017_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1017 gender: Male timepoint: post-surgery 1 library barcode: TCCGCGAA-CCTATCCT drug treatment: Placebo response group: Placebo | 831.0 |
| GSM3405484 | 1017_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1017 gender: Male timepoint: post-surgery 2 library barcode: TCTCGCGC-CCTATCCT drug treatment: Placebo response group: Placebo | 912.0 |
| GSM3405485 | 1018_baseline | cell type: peripheral blood mononuclear cell subject: 1018 gender: Female timepoint: baseline library barcode: AGCGATAG-CCTATCCT drug treatment: ibuprofen sodium response group: Full responder | 1613.0 |
| GSM3405486 | 1018_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1018 gender: Female timepoint: post-surgery 1 library barcode: ATTACTCG-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 1449.0 |
| GSM3405487 | 1018_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1018 gender: Female timepoint: post-surgery 2 library barcode: TCCGGAGA-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 1255.0 |
| GSM3405488 | 1019_baseline | cell type: peripheral blood mononuclear cell subject: 1019 gender: Male timepoint: baseline library barcode: CGCTCATT-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 1051.0 |
| GSM3405489 | 1019_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1019 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 1621.0 |
| GSM3405490 | 1019_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1019 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 864.0 |
| GSM3405491 | 1020_baseline | cell type: peripheral blood mononuclear cell subject: 1020 gender: Female timepoint: baseline library barcode: GAATTCGT-GGCTCTGA drug treatment: Placebo response group: Placebo | |
| GSM3405492 | 1020_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1020 gender: Female timepoint: post-surgery 1 library barcode: CTGAAGCT-GGCTCTGA drug treatment: Placebo response group: Placebo | 968.0 |
| GSM3405493 | 1020_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1020 gender: Female timepoint: post-surgery 2 library barcode: TAATGCGC-GGCTCTGA drug treatment: Placebo response group: Placebo | 1051.0 |
| GSM3405494 | 1021_baseline | cell type: peripheral blood mononuclear cell subject: 1021 gender: Female timepoint: baseline library barcode: CGGCTATG-GGCTCTGA drug treatment: ibuprofen sodium response group: Partial responder | 639.0 |
| GSM3405495 | 1021_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1021 gender: Female timepoint: post-surgery 1 library barcode: TCCGCGAA-GGCTCTGA drug treatment: ibuprofen sodium response group: Partial responder | 681.0 |
| GSM3405496 | 1021_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1021 gender: Female timepoint: post-surgery 2 library barcode: TCTCGCGC-GGCTCTGA drug treatment: ibuprofen sodium response group: Partial responder | 572.0 |
| GSM3405497 | 1022_baseline | cell type: peripheral blood mononuclear cell subject: 1022 gender: Male timepoint: baseline library barcode: AGCGATAG-GGCTCTGA drug treatment: ibuprofen sodium response group: Full responder | 1281.0 |
| GSM3405498 | 1022_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1022 gender: Male timepoint: post-surgery 1 library barcode: ATTACTCG-AGGCGAAG drug treatment: ibuprofen sodium response group: Full responder | 981.0 |
| GSM3405499 | 1022_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1022 gender: Male timepoint: post-surgery 2 library barcode: TCCGGAGA-AGGCGAAG drug treatment: ibuprofen sodium response group: Full responder | 1506.0 |
| GSM3405500 | 1023_baseline | cell type: peripheral blood mononuclear cell subject: 1023 gender: Male timepoint: baseline library barcode: CGCTCATT-AGGCGAAG drug treatment: Placebo response group: Placebo | 1150.0 |
| GSM3405501 | 1023_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1023 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-AGGCGAAG drug treatment: Placebo response group: Placebo | 800.0 |
| GSM3405502 | 1023_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1023 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-AGGCGAAG drug treatment: Placebo response group: Placebo | 715.0 |
| GSM3405503 | 1024_baseline | cell type: peripheral blood mononuclear cell subject: 1024 gender: Female timepoint: baseline library barcode: GAATTCGT-AGGCGAAG drug treatment: ibuprofen sodium response group: Partial responder | 1177.0 |
| GSM3405504 | 1024_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1024 gender: Female timepoint: post-surgery 1 library barcode: CTGAAGCT-AGGCGAAG drug treatment: ibuprofen sodium response group: Partial responder | 1271.0 |
| GSM3405505 | 1024_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1024 gender: Female timepoint: post-surgery 2 library barcode: TAATGCGC-AGGCGAAG drug treatment: ibuprofen sodium response group: Partial responder | 868.0 |
| GSM3405506 | 1026_baseline | cell type: peripheral blood mononuclear cell subject: 1026 gender: Female timepoint: baseline library barcode: CGGCTATG-AGGCGAAG drug treatment: Placebo response group: Placebo | 1013.0 |
| GSM3405507 | 1026_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1026 gender: Female timepoint: post-surgery 1 library barcode: TCCGCGAA-AGGCGAAG drug treatment: Placebo response group: Placebo | 950.0 |
| GSM3405508 | 1026_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1026 gender: Female timepoint: post-surgery 2 library barcode: TCTCGCGC-AGGCGAAG drug treatment: Placebo response group: Placebo | 833.0 |
| GSM3405509 | 1027_baseline | cell type: peripheral blood mononuclear cell subject: 1027 gender: Female timepoint: baseline library barcode: AGCGATAG-AGGCGAAG drug treatment: Placebo response group: Placebo | 1067.0 |
| GSM3405510 | 1027_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1027 gender: Female timepoint: post-surgery 1 library barcode: ATTACTCG-TAATCTTA drug treatment: Placebo response group: Placebo | 1014.0 |
| GSM3405511 | 1027_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1027 gender: Female timepoint: post-surgery 2 library barcode: TCCGGAGA-TAATCTTA drug treatment: Placebo response group: Placebo | 800.0 |
| GSM3405512 | 1028_baseline | cell type: peripheral blood mononuclear cell subject: 1028 gender: Male timepoint: baseline library barcode: CGCTCATT-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 871.0 |
| GSM3405513 | 1028_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1028 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 821.0 |
| GSM3405514 | 1028_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1028 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 862.0 |
| GSM3405515 | 1029_baseline | cell type: peripheral blood mononuclear cell subject: 1029 gender: Male timepoint: baseline library barcode: GAATTCGT-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 683.0 |
| GSM3405516 | 1029_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1029 gender: Male timepoint: post-surgery 1 library barcode: CTGAAGCT-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 499.0 |
| GSM3405517 | 1029_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1029 gender: Male timepoint: post-surgery 2 library barcode: TAATGCGC-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 526.0 |
| GSM3405518 | 1030_baseline | cell type: peripheral blood mononuclear cell subject: 1030 gender: Female timepoint: baseline library barcode: CGGCTATG-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 912.0 |
| GSM3405519 | 1030_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1030 gender: Female timepoint: post-surgery 1 library barcode: TCCGCGAA-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 985.0 |
| GSM3405520 | 1030_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1030 gender: Female timepoint: post-surgery 2 library barcode: TCTCGCGC-TAATCTTA drug treatment: ibuprofen sodium response group: Partial responder | 802.0 |
| GSM3405521 | 1032_baseline | cell type: peripheral blood mononuclear cell subject: 1032 gender: Female timepoint: baseline library barcode: AGCGATAG-TAATCTTA drug treatment: Placebo response group: Placebo | 1332.0 |
| GSM3405522 | 1032_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1032 gender: Female timepoint: post-surgery 1 library barcode: ATTACTCG-CAGGACGT drug treatment: Placebo response group: Placebo | 1285.0 |
| GSM3405523 | 1032_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1032 gender: Female timepoint: post-surgery 2 library barcode: TCCGGAGA-CAGGACGT drug treatment: Placebo response group: Placebo | 1250.0 |
| GSM3405524 | 1033_baseline | cell type: peripheral blood mononuclear cell subject: 1033 gender: Male timepoint: baseline library barcode: CGCTCATT-CAGGACGT drug treatment: ibuprofen sodium response group: Full responder | 966.0 |
| GSM3405525 | 1033_post-surgery_1 | cell type: peripheral blood mononuclear cell subject: 1033 gender: Male timepoint: post-surgery 1 library barcode: GAGATTCC-CAGGACGT drug treatment: ibuprofen sodium response group: Full responder | 1107.0 |
| GSM3405526 | 1033_post-surgery_2 | cell type: peripheral blood mononuclear cell subject: 1033 gender: Male timepoint: post-surgery 2 library barcode: ATTCAGAA-CAGGACGT drug treatment: ibuprofen sodium response group: Full responder | 990.0 |
| ID | GSE12860 |
| Title | Antirheumatic Drug Response in Human Chondrocytes: Potential Molecular Targets to Stimulate Cartilage Regeneration |
| Organism | Homo sapiens |
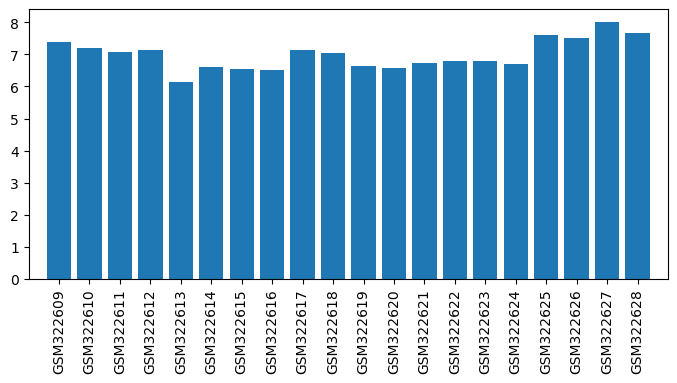
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM322609 | RASF stimulated chondrocytes rep 1 GSM253283 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium | 7.398045376000001 |
| GSM322610 | RASF stimulated chondrocytes rep 2 GSM253284 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium | 7.210532300000001 |
| GSM322611 | NDSF stimulated chondrocytes rep 1 GSM253285 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of NDSF cultured in the same medium | 7.068038091 |
| GSM322612 | NDSF stimulated chondrocytes rep 2 GSM253286 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of NDSF cultured in the same medium | 7.153831574 |
| GSM322613 | Chondrocytes stimulated with diclofenac treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with diclofenac for 48h | 6.153774473 |
| GSM322614 | Chondrocytes stimulated with diclofenac treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with diclofenac for 48h | 6.6160459215 |
| GSM322615 | Chondrocytes stimulated with prioxicam treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with piroxicam for 48h | 6.548832377 |
| GSM322616 | Chondrocytes stimulated with prioxicam treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with piroxicam for 48h | 6.529364022999999 |
| GSM322617 | Chondrocytes stimulated with chloroquine phosphate (resochin) treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with chloroquine phosphate (resochin) for 48h | 7.1542411585 |
| GSM322618 | Chondrocytes stimulated with chloroquine phosphate (resochin) treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with chloroquine phosphate (resochin) for 48h | 7.0312460495 |
| GSM322619 | Chondrocytes stimulated with sodium aurothiomalate (tauredon) treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with sodium aurothiomalate (tauredon) for 48h | 6.6470460515 |
| GSM322620 | Chondrocytes stimulated with sodium aurothiomalate (tauredon) treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with sodium aurothiomalate (tauredon) for 48h | 6.586736044 |
| GSM322621 | Chondrocytes stimulated with azathioprine (imurek) treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with azathioprine (imurek) for 48h | 6.727772334 |
| GSM322622 | Chondrocytes stimulated with azathioprine (imurek) treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with azathioprine (imurek) for 48h | 6.7816349215 |
| GSM322623 | Chondrocytes stimulated with methotrexate treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with methotrexate for 48h | 6.7836897405 |
| GSM322624 | Chondrocytes stimulated with methotrexate treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with methotrexate for 48h | 6.710672591 |
| GSM322625 | Chondrocytes stimulated with prednisolone (solu-decortin) treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with prednisolone (solu-decortin) for 48h | 7.6008382789999995 |
| GSM322626 | Chondrocytes stimulated with prednisolone (solu-decortin) treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with prednisolone (solu-decortin) for 48h | 7.530771192 |
| GSM322627 | Chondrocytes stimulated with methylprednisolone (urbason) treated RASF rep 1 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with methylprednisolone (urbason) for 48h | 8.019646401 |
| GSM322628 | Chondrocytes stimulated with methylprednisolone (urbason) treated RASF rep 2 | Human chondrocytes cultured in RPMI 1640 medium supplemented with 10% human serum, 100ng/ml amphotericin B, 100U/ml penicillin and 100µg/ml streptomycin for 2 passages, 3D cultured for 14 days in alginate beads in presence of 170µM L-ascorbic acid 2-phosphate and subsequently stimulated for 48h with supernatant of RASF cultured in the same medium and treated with methylprednisolone (urbason) for 48h | 7.6845993855 |
| ID | GSE150408 |
| Title | Identification of key genes potentially related to intervertebral disc degeneration by microarray analysis |
| Organism | Homo sapiens |
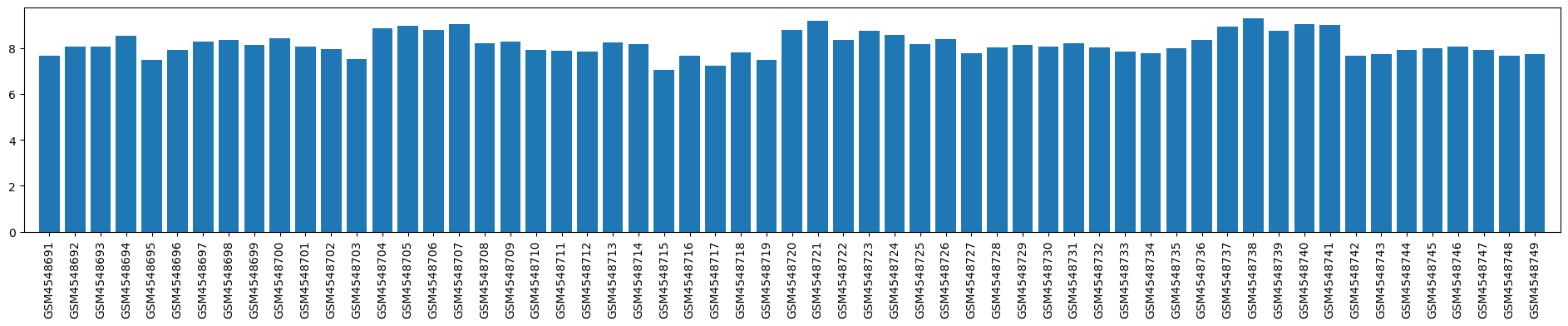
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM4548691 | volunteer-9 | tissue: whole blood diagnosis: healthy control treatment: n/a | 7.685837816 |
| GSM4548692 | volunteer-10 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.054875032 |
| GSM4548693 | volunteer-11 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.052060401 |
| GSM4548694 | volunteer-12 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.542047585 |
| GSM4548695 | volunteer-13 | tissue: whole blood diagnosis: healthy control treatment: n/a | 7.503834167 |
| GSM4548696 | volunteer-14 | tissue: whole blood diagnosis: healthy control treatment: n/a | 7.918809559 |
| GSM4548697 | volunteer-15 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.304095094 |
| GSM4548698 | volunteer-16 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.368947185 |
| GSM4548699 | volunteer-17 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.135736644 |
| GSM4548700 | volunteer-18 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.422538201 |
| GSM4548701 | volunteer-19 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.085101524 |
| GSM4548702 | volunteer-20 | tissue: whole blood diagnosis: healthy control treatment: n/a | 7.970276551 |
| GSM4548703 | volunteer-21 | tissue: whole blood diagnosis: healthy control treatment: n/a | 7.523029894 |
| GSM4548704 | volunteer-22 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.87039137 |
| GSM4548705 | volunteer-23 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.958587023 |
| GSM4548706 | volunteer-24 | tissue: whole blood diagnosis: healthy control treatment: n/a | 8.782315886 |
| GSM4548707 | volunteer-25 | tissue: whole blood diagnosis: healthy control treatment: n/a | 9.034115821 |
| GSM4548708 | patient-9 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.220053625 |
| GSM4548709 | patient-10 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.303079203 |
| GSM4548710 | patient-11 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.937511477 |
| GSM4548711 | patient-12 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.893960416 |
| GSM4548712 | patient-13 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.843854157 |
| GSM4548713 | patient-14 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.248255417 |
| GSM4548714 | patient-15 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.163212949 |
| GSM4548715 | patient-16 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.053120429 |
| GSM4548716 | patient-17 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.670500418 |
| GSM4548717 | patient-18 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.238413793 |
| GSM4548718 | patient-19 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.806090738 |
| GSM4548719 | patient-20 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 7.482179789 |
| GSM4548720 | patient-21 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.809342509 |
| GSM4548721 | patient-22 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 9.184718869 |
| GSM4548722 | patient-23 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.343931048 |
| GSM4548723 | patient-24 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.761176757 |
| GSM4548724 | patient-25 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: none | 8.560324885 |
| GSM4548725 | treatment-1 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.170983611 |
| GSM4548726 | treatment-2 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.396951774 |
| GSM4548727 | treatment-3 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.785185668 |
| GSM4548728 | treatment-4 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.016097289 |
| GSM4548729 | treatment-5 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.146813965 |
| GSM4548730 | treatment-6 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.085873736 |
| GSM4548731 | treatment-7 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.215835935 |
| GSM4548732 | treatment-8 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.041389356 |
| GSM4548733 | treatment-9 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.842693987 |
| GSM4548734 | treatment-10 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.791194921 |
| GSM4548735 | treatment-11 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.008899703 |
| GSM4548736 | treatment-12 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.373343894 |
| GSM4548737 | treatment-13 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.954993672 |
| GSM4548738 | treatment-14 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 9.301252835 |
| GSM4548739 | treatment-15 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.760172053 |
| GSM4548740 | treatment-16 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 9.050285186 |
| GSM4548741 | treatment-17 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 9.028497262 |
| GSM4548742 | treatment-18 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.655676246 |
| GSM4548743 | treatment-19 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.745528156 |
| GSM4548744 | treatment-20 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.934859388 |
| GSM4548745 | treatment-21 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.99419788 |
| GSM4548746 | treatment-22 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 8.077395593 |
| GSM4548747 | treatment-23 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.910736848 |
| GSM4548748 | treatment-24 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.683670033 |
| GSM4548749 | treatment-25 | tissue: whole blood diagnosis: intervertebral disc degeneration treatment: Traditional Chinese Medicine | 7.748876386 |
| ID | GSE166193 |
| Title | Expression data from human astrocytes (HA) under oxygen-glucose deprivation / reoxygenation |
| Organism | Homo sapiens |
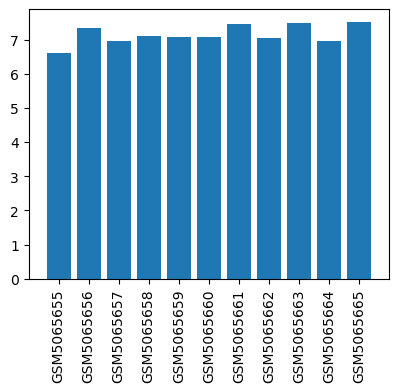
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM5065655 | without DEX pretreatment, reoxygenation time is 0 h | cell type: astrocyte treatment: without DEX pretreatment and reoxygenated for 0 h tissue: brain | 6.5988 |
| GSM5065656 | without DEX pretreatment, reoxygenation time is 2 h | cell type: astrocyte treatment: without DEX pretreatment and reoxygenated for 2 h tissue: brain | 7.35002 |
| GSM5065657 | without DEX pretreatment, reoxygenation time is 6 h | cell type: astrocyte treatment: without DEX pretreatment and reoxygenated for 6 h tissue: brain | 6.97214 |
| GSM5065658 | without DEX pretreatment, reoxygenation time is 12 h | cell type: astrocyte treatment: without DEX pretreatment and reoxygenated for 12 h tissue: brain | 7.10746 |
| GSM5065659 | without DEX pretreatment, reoxygenation time is 24 h | cell type: astrocyte treatment: without DEX pretreatment and reoxygenated for 24 h tissue: brain | 7.08723 |
| GSM5065660 | with DEX pretreatment, reoxygenation time is 0 h | cell type: astrocyte treatment: with DEX pretreatment and reoxygenated for 0 h tissue: brain | 7.08104 |
| GSM5065661 | with DEX pretreatment, reoxygenation time is 2 h | cell type: astrocyte treatment: with DEX pretreatment and reoxygenated for 2 h tissue: brain | 7.47044 |
| GSM5065662 | with DEX pretreatment, reoxygenation time is 6 h | cell type: astrocyte treatment: with DEX pretreatment and reoxygenated for 6 h tissue: brain | 7.04413 |
| GSM5065663 | with DEX pretreatment, reoxygenation time is 12 h | cell type: astrocyte treatment: with DEX pretreatment and reoxygenated for 12 h tissue: brain | 7.4796 |
| GSM5065664 | with DEX pretreatment, reoxygenation time is 24 h | cell type: astrocyte treatment: with DEX pretreatment and reoxygenated for 24 h tissue: brain | 6.97199 |
| GSM5065665 | control | cell type: astrocyte treatment: without stress tissue: brain | 7.51668 |
| ID | GSE179004 |
| Title | Expression data from glioblastoma surgical specimens |
| Organism | Homo sapiens |
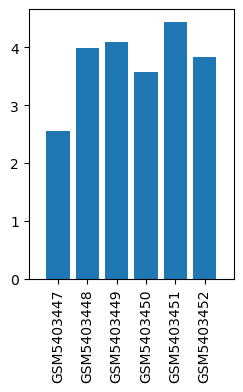
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM5403447 | glioblastoma tissue 1 in TIVA group | tissue: glioblastoma | 2.552403622 |
| GSM5403448 | glioblastoma tissue 2 in TIVA group | tissue: glioblastoma | 3.9877110499999997 |
| GSM5403449 | glioblastoma tissue 3 in TIVA group | tissue: glioblastoma | 4.0862203945 |
| GSM5403450 | glioblastoma tissue 1 in INHA group | tissue: glioblastoma | 3.5779253925 |
| GSM5403451 | glioblastoma tissue 2 in INHA group | tissue: glioblastoma | 4.4383383 |
| GSM5403452 | glioblastoma tissue 3 in INHA group | tissue: glioblastoma | 3.839997645 |
| ID | GSE212044 |
| Title | Reversibility of etomidate-induced contractile arrest through activation of myocardial autophagy |
| Organism | Homo sapiens |
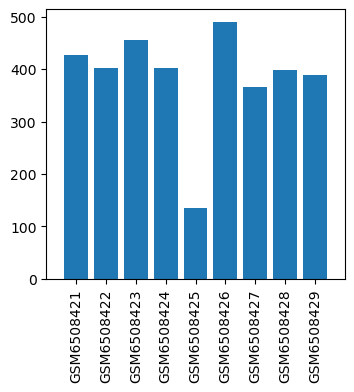
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM6508421 | IPSC-CM cells, DSMO 1 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: DSMO time: Day 30 | 427.0 |
| GSM6508422 | IPSC-CM cells, DSMO 2 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: DSMO time: Day 30 | 402.0 |
| GSM6508423 | IPSC-CM cells, DSMO 3 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: DSMO time: Day 30 | 455.0 |
| GSM6508424 | IPSC-CM cells, Etomidate 1 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Etomidate time: Day 30 | 402.0 |
| GSM6508425 | IPSC-CM cells, Etomidate 2 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Etomidate time: Day 30 | 134.0 |
| GSM6508426 | IPSC-CM cells, Etomidate 3 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Etomidate time: Day 30 | 490.0 |
| GSM6508427 | IPSC-CM cells, Propofol 1 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Propofol time: Day 30 | 366.0 |
| GSM6508428 | IPSC-CM cells, Propofol 2 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Propofol time: Day 30 | 398.0 |
| GSM6508429 | IPSC-CM cells, Propofol 3 | cell line: Induced pluripotent stem cells (IPSC) cell type: iPSC-derived Cardiomyocytes(IPSC-CM) phenotype: dilated (DCM) disease state: Duchenne Muscular Dystrophy (DMD) treatment: Propofol time: Day 30 | 389.0 |
| ID | GSE255952 |
| Title | Transcriptome data of B cells and T helper cells from patients with multiple sclerosis receiving methylprednisolone for the treatment of an acute relapse |
| Organism | Homo sapiens |
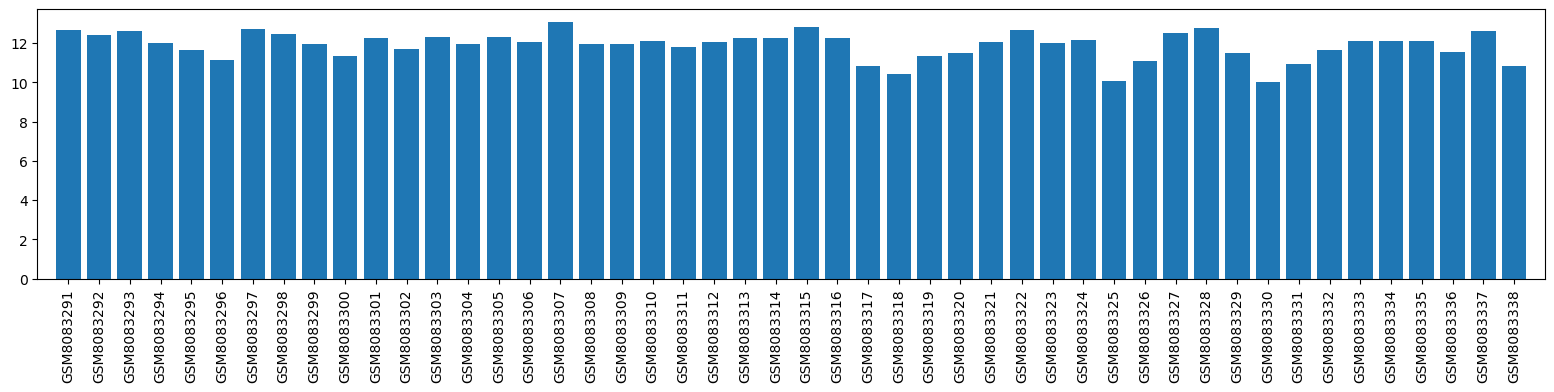
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM8083291 | patient C1P1, before first course of MP, T cells | tissue: peripheral blood patient id: C1P1 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 34.9 cells per ml: 2982000 purity (%): 98.1 rna integrity number: 7.8 age (years): 60 Sex: female body weight (in kg): 75.4 body size (in cm): 168 body mass index: 26.7 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 11 number of relapses in the past 2 years: 0 duration since last relapse (months): 133 edss score: 3.5 disease-modifying therapy: teriflunomide number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: hair loss | 12.67503 |
| GSM8083292 | patient C1P1, after first course of MP, T cells | tissue: peripheral blood patient id: C1P1 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 25.9 cells per ml: 304500 purity (%): 95.3 rna integrity number: 8.7 age (years): 60 Sex: female body weight (in kg): 75.4 body size (in cm): 168 body mass index: 26.7 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 11 number of relapses in the past 2 years: 0 duration since last relapse (months): 133 edss score: NA disease-modifying therapy: teriflunomide number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: worse fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: hair loss | 12.38785 |
| GSM8083293 | patient C1P1, before second course of MP, T cells | tissue: peripheral blood patient id: C1P1 mp course: second time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 27.8 cells per ml: 1254750 purity (%): 96.7 rna integrity number: 8.3 age (years): 60 Sex: female body weight (in kg): 75.4 body size (in cm): 168 body mass index: 26.7 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 11 number of relapses in the past 2 years: 0 duration since last relapse (months): 134 edss score: 3.5 disease-modifying therapy: teriflunomide number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 6000 days from first to last mp application: 2 side effects to mp: headache, diarrhea | 12.63022 |
| GSM8083294 | patient C1P1, after second course of MP, T cells | tissue: peripheral blood patient id: C1P1 mp course: second time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 6.6 cells per ml: 231000 purity (%): 93.7 rna integrity number: 9.4 age (years): 60 Sex: female body weight (in kg): 75.4 body size (in cm): 168 body mass index: 26.7 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 11 number of relapses in the past 2 years: 0 duration since last relapse (months): 134 edss score: NA disease-modifying therapy: teriflunomide number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: worse fs: sensory: better fs: visual: unchanged total dose of mp in mg: 6000 days from first to last mp application: 2 side effects to mp: headache, diarrhea | 12.00121 |
| GSM8083295 | patient C1P2, before first course of MP, B cells | tissue: peripheral blood patient id: C1P2 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 4.7 cells per ml: 236250 purity (%): 91.9 rna integrity number: 9.0 age (years): 38 Sex: male body weight (in kg): 67 body size (in cm): 183 body mass index: 20.0 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 17 number of relapses in the past 2 years: 0 duration since last relapse (months): 206 edss score: 3.0 disease-modifying therapy: teriflunomide number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: yes fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 11.62039 |
| GSM8083296 | patient C1P2, after first course of MP, B cells | tissue: peripheral blood patient id: C1P2 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 3.7 cells per ml: 283500 purity (%): 91.0 rna integrity number: 9.1 age (years): 38 Sex: male body weight (in kg): 67 body size (in cm): 183 body mass index: 20.0 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 17 number of relapses in the past 2 years: 0 duration since last relapse (months): 206 edss score: 3.0 disease-modifying therapy: teriflunomide number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: better fs: cerebral: unchanged fs: pyramidal: better fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 11.13209 |
| GSM8083297 | patient C1P2, before first course of MP, T cells | tissue: peripheral blood patient id: C1P2 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 28.7 cells per ml: 693000 purity (%): 96.0 rna integrity number: 8.3 age (years): 38 Sex: male body weight (in kg): 67 body size (in cm): 183 body mass index: 20.0 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 17 number of relapses in the past 2 years: 0 duration since last relapse (months): 206 edss score: 3.0 disease-modifying therapy: teriflunomide number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: yes fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 12.69237 |
| GSM8083298 | patient C1P2, after first course of MP, T cells | tissue: peripheral blood patient id: C1P2 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 11.5 cells per ml: 624750 purity (%): 96.0 rna integrity number: 8.3 age (years): 38 Sex: male body weight (in kg): 67 body size (in cm): 183 body mass index: 20.0 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 17 number of relapses in the past 2 years: 0 duration since last relapse (months): 206 edss score: 3.0 disease-modifying therapy: teriflunomide number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: better fs: cerebral: unchanged fs: pyramidal: better fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 12.47845 |
| GSM8083299 | patient C1P3, before first course of MP, B cells | tissue: peripheral blood patient id: C1P3 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 10.0 cells per ml: 498750 purity (%): 98.8 rna integrity number: 8.9 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 11.93943 |
| GSM8083300 | patient C1P3, after first course of MP, B cells | tissue: peripheral blood patient id: C1P3 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 6.7 cells per ml: 383250 purity (%): 97.9 rna integrity number: 9.4 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 11.34448 |
| GSM8083301 | patient C1P3, before first course of MP, T cells | tissue: peripheral blood patient id: C1P3 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 31.0 cells per ml: 1134000 purity (%): 94.7 rna integrity number: 8.1 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 12.26694 |
| GSM8083302 | patient C1P3, after first course of MP, T cells | tissue: peripheral blood patient id: C1P3 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 18.1 cells per ml: 640500 purity (%): 96.2 rna integrity number: 8.1 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 11.68604 |
| GSM8083303 | patient C1P3, before second course of MP, T cells | tissue: peripheral blood patient id: C1P3 mp course: second time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 30.3 cells per ml: 897750 purity (%): 97.0 rna integrity number: 8.5 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 6000 days from first to last mp | 12.2834 |
| GSM8083304 | patient C1P3, after second course of MP, T cells | tissue: peripheral blood patient id: C1P3 mp course: second time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 27.7 cells per ml: 1433250 purity (%): 97.5 rna integrity number: 8.3 age (years): 34 Sex: female body weight (in kg): 63 body size (in cm): 168 body mass index: 22.3 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 8 number of relapses in the past 2 years: 1 duration since last relapse (months): 5 edss score: 2.5 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 1 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 6000 days from first to last mp application: 5 side effects to mp: none | 11.96379 |
| GSM8083305 | patient C1P4, before first course of MP, B cells | tissue: peripheral blood patient id: C1P4 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 7.3 cells per ml: 378000 purity (%): 96.3 rna integrity number: 9.6 age (years): 53 Sex: female body weight (in kg): 58 body size (in cm): 159 body mass index: 22.9 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 3 number of relapses in the past 2 years: 0 duration since last relapse (months): 34 edss score: 3.0 disease-modifying therapy: glatiramer acetate number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 12.27989 |
| GSM8083306 | patient C1P4, after first course of MP, B cells | tissue: peripheral blood patient id: C1P4 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 7.5 cells per ml: 939750 purity (%): 96.7 rna integrity number: 8.8 age (years): 53 Sex: female body weight (in kg): 58 body size (in cm): 159 body mass index: 22.9 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 3 number of relapses in the past 2 years: 0 duration since last relapse (months): 34 edss score: NA disease-modifying therapy: glatiramer acetate number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 12.0564 |
| GSM8083307 | patient C1P4, before first course of MP, T cells | tissue: peripheral blood patient id: C1P4 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 38.1 cells per ml: 1522500 purity (%): 98.7 rna integrity number: NA age (years): 53 Sex: female body weight (in kg): 58 body size (in cm): 159 body mass index: 22.9 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 3 number of relapses in the past 2 years: 0 duration since last relapse (months): 34 edss score: 3.0 disease-modifying therapy: glatiramer acetate number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 13.06978 |
| GSM8083308 | patient C1P4, after first course of MP, T cells | tissue: peripheral blood patient id: C1P4 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 15.9 cells per ml: 1617000 purity (%): 98.3 rna integrity number: 8.5 age (years): 53 Sex: female body weight (in kg): 58 body size (in cm): 159 body mass index: 22.9 current smoker: no study center: Berlin course of disease: CIS/RRMS disease duration (years): 3 number of relapses in the past 2 years: 0 duration since last relapse (months): 34 edss score: NA disease-modifying therapy: glatiramer acetate number of other medications taken: 5 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 11.92632 |
| GSM8083309 | patient C1P5, before first course of MP, T cells | tissue: peripheral blood patient id: C1P5 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 31.6 cells per ml: 1344000 purity (%): 99.1 rna integrity number: 8.0 age (years): 42 Sex: male body weight (in kg): 57 body size (in cm): 180 body mass index: 17.6 current smoker: yes study center: Berlin course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 7.5 disease-modifying therapy: ocrelizumab number of other medications taken: 6 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: no fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 5 side effects to mp: none | 11.95233 |
| GSM8083310 | patient C1P5, after first course of MP, T cells | tissue: peripheral blood patient id: C1P5 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 21.9 cells per ml: 1165500 purity (%): 98.9 rna integrity number: 8.4 age (years): 42 Sex: male body weight (in kg): 57 body size (in cm): 180 body mass index: 17.6 current smoker: yes study center: Berlin course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 7.5 disease-modifying therapy: ocrelizumab number of other medications taken: 6 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 5 side effects to mp: none | 12.10918 |
| GSM8083311 | patient C1P5, before second course of MP, T cells | tissue: peripheral blood patient id: C1P5 mp course: second time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 26.9 cells per ml: 1233750 purity (%): 97.8 rna integrity number: 8.5 age (years): 42 Sex: male body weight (in kg): 57 body size (in cm): 180 body mass index: 17.6 current smoker: yes study center: Berlin course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 7.5 disease-modifying therapy: ocrelizumab number of other medications taken: 6 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: no fs: visual: no total dose of mp in mg: 4000 days from first to last mp application: 2 side effects to mp: cardiovascular problems | 11.79063 |
| GSM8083312 | patient C1P5, after second course of MP, T cells | tissue: peripheral blood patient id: C1P5 mp course: second time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 11.0 cells per ml: 514500 purity (%): 98.6 rna integrity number: 8.7 age (years): 42 Sex: male body weight (in kg): 57 body size (in cm): 180 body mass index: 17.6 current smoker: yes study center: Berlin course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 7.5 disease-modifying therapy: ocrelizumab number of other medications taken: 6 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 4000 days from first to last mp application: 2 side effects to mp: cardiovascular problems | 12.06746 |
| GSM8083313 | patient C2P1, before first course of MP, B cells | tissue: peripheral blood patient id: C2P1 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 11.7 cells per ml: 2604000 purity (%): 96.7 rna integrity number: 8.5 age (years): 24 Sex: female body weight (in kg): 60 body size (in cm): 170 body mass index: 20.8 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 6 edss score: 3.0 disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.26746 |
| GSM8083314 | patient C2P1, after first course of MP, B cells | tissue: peripheral blood patient id: C2P1 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 8.8 cells per ml: 630000 purity (%): 97.4 rna integrity number: 8.4 age (years): 24 Sex: female body weight (in kg): 60 body size (in cm): 170 body mass index: 20.8 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 6 edss score: NA disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.26946 |
| GSM8083315 | patient C2P1, before first course of MP, T cells | tissue: peripheral blood patient id: C2P1 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 41.5 cells per ml: 2436000 purity (%): 94.6 rna integrity number: 8.0 age (years): 24 Sex: female body weight (in kg): 60 body size (in cm): 170 body mass index: 20.8 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 6 edss score: 3.0 disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.80123 |
| GSM8083316 | patient C2P1, after first course of MP, T cells | tissue: peripheral blood patient id: C2P1 mp course: first time point: after cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 6.2 cells per ml: 346500 purity (%): 86.5 rna integrity number: 8.0 age (years): 24 Sex: female body weight (in kg): 60 body size (in cm): 170 body mass index: 20.8 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 6 edss score: NA disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.24361 |
| GSM8083317 | patient C2P2, before first course of MP, B cells | tissue: peripheral blood patient id: C2P2 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 0.9 cells per ml: 467250 purity (%): 87.3 rna integrity number: 8.8 age (years): 41 Sex: female body weight (in kg): 71 body size (in cm): 170 body mass index: 24.6 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 3 duration since last relapse (months): 3 edss score: NA disease-modifying therapy: glatiramer acetate number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 10.83024 |
| GSM8083318 | patient C2P2, after first course of MP, B cells | tissue: peripheral blood patient id: C2P2 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 6.3 cells per ml: 199500 purity (%): 97.6 rna integrity number: 9.5 age (years): 41 Sex: female body weight (in kg): 71 body size (in cm): 170 body mass index: 24.6 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 3 duration since last relapse (months): 3 edss score: 1.5 disease-modifying therapy: glatiramer acetate number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 10.43142 |
| GSM8083319 | patient C2P3, before first course of MP, B cells | tissue: peripheral blood patient id: C2P3 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 12.2 cells per ml: 1170750 purity (%): 97.2 rna integrity number: 8.5 age (years): 20 Sex: female body weight (in kg): 55 body size (in cm): 165 body mass index: 20.2 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 6 number of relapses in the past 2 years: 0 duration since last relapse (months): NA edss score: 1.5 disease-modifying therapy: natalizumab number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 11.36261 |
| GSM8083320 | patient C2P3, after first course of MP, B cells | tissue: peripheral blood patient id: C2P3 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 12.2 cells per ml: 1170750 purity (%): 97.2 rna integrity number: 8.5 age (years): 20 Sex: female body weight (in kg): 55 body size (in cm): 165 body mass index: 20.2 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 6 number of relapses in the past 2 years: 0 duration since last relapse (months): NA edss score: 1.5 disease-modifying therapy: natalizumab number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 11.5105 |
| GSM8083321 | patient C2P3, before first course of MP, T cells | tissue: peripheral blood patient id: C2P3 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 20.2 cells per ml: 1711500 purity (%): 94.3 rna integrity number: 7.3 age (years): 20 Sex: female body weight (in kg): 55 body size (in cm): 165 body mass index: 20.2 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 6 number of relapses in the past 2 years: 0 duration since last relapse (months): NA edss score: 1.5 disease-modifying therapy: natalizumab number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.02561 |
| GSM8083322 | patient C2P3, after first course of MP, T cells | tissue: peripheral blood patient id: C2P3 mp course: first time point: before cell population: CD4+ T cells clinical evaluation: responder percentage in pbmc: 22.8 cells per ml: 992250 purity (%): 96.5 rna integrity number: 8.2 age (years): 20 Sex: female body weight (in kg): 55 body size (in cm): 165 body mass index: 20.2 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 6 number of relapses in the past 2 years: 0 duration since last relapse (months): NA edss score: 1.5 disease-modifying therapy: natalizumab number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.66411 |
| GSM8083323 | patient C2P4, before first course of MP, B cells | tissue: peripheral blood patient id: C2P4 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 8.0 cells per ml: 1690500 purity (%): 96.3 rna integrity number: 9.0 age (years): 60 Sex: male body weight (in kg): 97 body size (in cm): 170 body mass index: 33.6 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 7 number of relapses in the past 2 years: 1 duration since last relapse (months): 10 edss score: 2.0 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 8 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: yes fs: pyramidal: no fs: sensory: no fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.01005 |
| GSM8083324 | patient C2P4, after first course of MP, B cells | tissue: peripheral blood patient id: C2P4 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 2.2 cells per ml: 273000 purity (%): 93.0 rna integrity number: 8.3 age (years): 60 Sex: male body weight (in kg): 97 body size (in cm): 170 body mass index: 33.6 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 7 number of relapses in the past 2 years: 1 duration since last relapse (months): 10 edss score: 2.0 disease-modifying therapy: peginterferon beta-1a number of other medications taken: 8 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.1409 |
| GSM8083325 | patient C2P5, before first course of MP, B cells | tissue: peripheral blood patient id: C2P5 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 2.6 cells per ml: 136500 purity (%): 53.8 rna integrity number: 6.9 age (years): 59 Sex: female body weight (in kg): 60 body size (in cm): 172 body mass index: 20.3 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 4 number of relapses in the past 2 years: 1 duration since last relapse (months): 22 edss score: 3.0 disease-modifying therapy: none number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 10.07574 |
| GSM8083326 | patient C2P5, after first course of MP, B cells | tissue: peripheral blood patient id: C2P5 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 6.8 cells per ml: 687750 purity (%): 98.1 rna integrity number: 8.2 age (years): 59 Sex: female body weight (in kg): 60 body size (in cm): 172 body mass index: 20.3 current smoker: yes study center: Greifswald course of disease: CIS/RRMS disease duration (years): 4 number of relapses in the past 2 years: 1 duration since last relapse (months): 22 edss score: 2.0 disease-modifying therapy: none number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: better fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 4 side effects to mp: none | 11.09091 |
| GSM8083327 | patient C2P6, before first course of MP, B cells | tissue: peripheral blood patient id: C2P6 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 4.2 cells per ml: 546000 purity (%): 92.4 rna integrity number: 8.8 age (years): 39 Sex: female body weight (in kg): 54 body size (in cm): 164 body mass index: 20.1 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 18 number of relapses in the past 2 years: 0 duration since last relapse (months): 47 edss score: 6.5 disease-modifying therapy: dimethyl fumarate number of other medications taken: 3 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: no fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.48804 |
| GSM8083328 | patient C2P6, after first course of MP, B cells | tissue: peripheral blood patient id: C2P6 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 6.6 cells per ml: 714000 purity (%): 97.8 rna integrity number: 9.0 age (years): 39 Sex: female body weight (in kg): 54 body size (in cm): 164 body mass index: 20.1 current smoker: no study center: Greifswald course of disease: CIS/RRMS disease duration (years): 18 number of relapses in the past 2 years: 0 duration since last relapse (months): 47 edss score: NA disease-modifying therapy: dimethyl fumarate number of other medications taken: 3 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 12.74572 |
| GSM8083329 | patient C2P7, before first course of MP, B cells | tissue: peripheral blood patient id: C2P7 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 2.7 cells per ml: 215250 purity (%): 95.6 rna integrity number: 7.8 age (years): 36 Sex: male body weight (in kg): 90 body size (in cm): NA body mass index: NA current smoker: NA study center: Greifswald course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 1 duration since last relapse (months): 21 edss score: 3.0 disease-modifying therapy: none number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: yes fs: cerebellar: no fs: cerebral: yes fs: pyramidal: no fs: sensory: no fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 2 side effects to mp: none | 11.51459 |
| GSM8083330 | patient C2P7, after first course of MP, B cells | tissue: peripheral blood patient id: C2P7 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 2.8 cells per ml: 162750 purity (%): 93.2 rna integrity number: 7.8 age (years): 36 Sex: male body weight (in kg): 90 body size (in cm): NA body mass index: NA current smoker: NA study center: Greifswald course of disease: CIS/RRMS disease duration (years): 16 number of relapses in the past 2 years: 1 duration since last relapse (months): 21 edss score: 3.0 disease-modifying therapy: none number of other medications taken: 1 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: better fs: pyramidal: unchanged fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 2 side effects to mp: none | 10.00694 |
| GSM8083331 | patient C2P8, before first course of MP, B cells | tissue: peripheral blood patient id: C2P8 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 3.5 cells per ml: 220500 purity (%): 95.5 rna integrity number: 8.5 age (years): 23 Sex: female body weight (in kg): NA body size (in cm): NA body mass index: NA current smoker: NA study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 10 edss score: 2.0 disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: no total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 10.91271 |
| GSM8083332 | patient C2P8, after first course of MP, B cells | tissue: peripheral blood patient id: C2P8 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: non-responder percentage in pbmc: 11.0 cells per ml: 535500 purity (%): 97.3 rna integrity number: 7.5 age (years): 23 Sex: female body weight (in kg): NA body size (in cm): NA body mass index: NA current smoker: NA study center: Greifswald course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 1 duration since last relapse (months): 10 edss score: NA disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 3000 days from first to last mp application: 2 side effects to mp: none | 11.66328 |
| GSM8083333 | patient C3P1, before second course of MP, T cells | tissue: peripheral blood patient id: C3P1 mp course: second time point: before cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 24.0 cells per ml: 225750 purity (%): 97.0 rna integrity number: 7.9 age (years): 36 Sex: female body weight (in kg): 63 body size (in cm): 175 body mass index: 20.6 current smoker: yes study center: Jena course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 2.5 disease-modifying therapy: dimethyl fumarate number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: no fs: sensory: yes fs: visual: yes total dose of mp in mg: 10000 days from first to last mp application: 6 side effects to mp: none | 12.11041 |
| GSM8083334 | patient C3P1, after second course of MP, T cells | tissue: peripheral blood patient id: C3P1 mp course: second time point: after cell population: CD4+ T cells clinical evaluation: non-responder percentage in pbmc: 10.1 cells per ml: 315000 purity (%): 98.6 rna integrity number: 8.3 age (years): 36 Sex: female body weight (in kg): 63 body size (in cm): 175 body mass index: 20.6 current smoker: yes study center: Jena course of disease: CIS/RRMS disease duration (years): 0 number of relapses in the past 2 years: 2 duration since last relapse (months): NA edss score: 2.5 disease-modifying therapy: dimethyl fumarate number of other medications taken: 0 neurological dysfunction: recurring fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: unchanged fs: sensory: better fs: visual: worse total dose of mp in mg: 10000 days from first to last mp application: 6 side effects to mp: none | 12.12005 |
| GSM8083335 | patient C4P1, before first course of MP, B cells | tissue: peripheral blood patient id: C4P1 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 5.3 cells per ml: 336000 purity (%): 96.3 rna integrity number: 9.4 age (years): 45 Sex: male body weight (in kg): 90 body size (in cm): 180 body mass index: 27.8 current smoker: yes study center: Rostock course of disease: CIS/RRMS disease duration (years): 13 number of relapses in the past 2 years: 0 duration since last relapse (months): 166 edss score: 3.5 disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: no fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: yes fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 5 side effects to mp: hyperhidrosis | 12.11993 |
| GSM8083336 | patient C4P1, after first course of MP, B cells | tissue: peripheral blood patient id: C4P1 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 6.9 cells per ml: 1417500 purity (%): 96.1 rna integrity number: 8.7 age (years): 45 Sex: male body weight (in kg): 90 body size (in cm): 180 body mass index: 27.8 current smoker: yes study center: Rostock course of disease: CIS/RRMS disease duration (years): 13 number of relapses in the past 2 years: 0 duration since last relapse (months): 166 edss score: 3.5 disease-modifying therapy: none number of other medications taken: 0 neurological dysfunction: new fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 5 side effects to mp: hyperhidrosis | 11.56191 |
| GSM8083337 | patient C4P2, before first course of MP, B cells | tissue: peripheral blood patient id: C4P2 mp course: first time point: before cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: NA cells per ml: 493500 purity (%): 94.2 rna integrity number: 8.2 age (years): 54 Sex: female body weight (in kg): 93 body size (in cm): 173 body mass index: 31.1 current smoker: no study center: Rostock course of disease: SPMS disease duration (years): 20 number of relapses in the past 2 years: 1 duration since last relapse (months): 11 edss score: 7.5 disease-modifying therapy: none number of other medications taken: 3 neurological dysfunction: NA fs: bladder or bowel: yes fs: brainstem: no fs: cerebellar: no fs: cerebral: no fs: pyramidal: yes fs: sensory: no fs: visual: no total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 12.60277 |
| GSM8083338 | patient C4P2, after first course of MP, B cells | tissue: peripheral blood patient id: C4P2 mp course: first time point: after cell population: CD19+ B cells clinical evaluation: responder percentage in pbmc: 0.4 cells per ml: 236250 purity (%): 42.7 rna integrity number: 8.1 age (years): 54 Sex: female body weight (in kg): 93 body size (in cm): 173 body mass index: 31.1 current smoker: no study center: Rostock course of disease: SPMS disease duration (years): 20 number of relapses in the past 2 years: 1 duration since last relapse (months): 11 edss score: 6.5 disease-modifying therapy: none number of other medications taken: 3 neurological dysfunction: NA fs: bladder or bowel: unchanged fs: brainstem: unchanged fs: cerebellar: unchanged fs: cerebral: unchanged fs: pyramidal: better fs: sensory: unchanged fs: visual: unchanged total dose of mp in mg: 5000 days from first to last mp application: 6 side effects to mp: none | 10.80861 |
| ID | GSE4386 |
| Title | Transcriptomics in cardiac surgery |
| Organism | Homo sapiens |
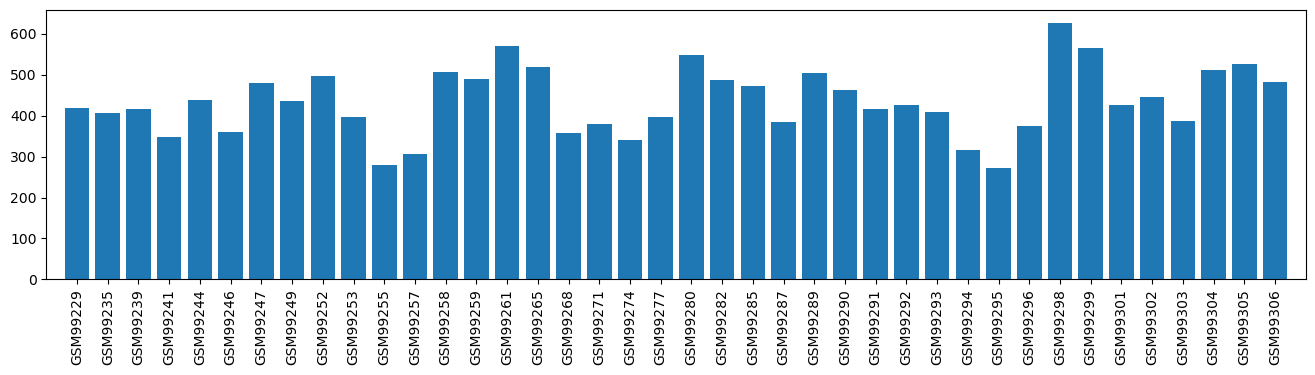
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM99229 | H01_S_B1 | male CABG patient, atrial tissue baseline before CABG surgery | 419.6899458563333 |
| GSM99235 | H01_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 406.49547674033334 |
| GSM99239 | H02_P_B1 | male CABG patient, atrial tissue baseline before CABG surgery | 415.3280827556667 |
| GSM99241 | H02_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 347.2628255486666 |
| GSM99244 | H03_P_B1 | male CABG patient, atrial tissue baseline before CABG surgery | 437.203001955 |
| GSM99246 | H03_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 359.17754492599994 |
| GSM99247 | H04_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 480.47056651233333 |
| GSM99249 | H04_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 436.4306562046666 |
| GSM99252 | H05_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 497.52046048133326 |
| GSM99253 | H05_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 397.458526296 |
| GSM99255 | H06_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 278.3290125426667 |
| GSM99257 | H06_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 306.42304769833333 |
| GSM99258 | H07_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 507.4709398336667 |
| GSM99259 | H07_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 489.1519658923333 |
| GSM99261 | H08_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 570.5510369963333 |
| GSM99265 | H08_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 519.82668516 |
| GSM99268 | H09_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 358.04433855300005 |
| GSM99271 | H09_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 379.2667486193334 |
| GSM99274 | H10_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 339.587164984 |
| GSM99277 | H10_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 396.409243969 |
| GSM99280 | H11_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 548.4145354590001 |
| GSM99282 | H11_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 487.73978082499997 |
| GSM99285 | H12_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 473.4239625356666 |
| GSM99287 | H12_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 383.3869081806667 |
| GSM99289 | H13_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 504.1344104976667 |
| GSM99290 | H13_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 462.68812413866675 |
| GSM99291 | H14_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 417.42735666199997 |
| GSM99292 | H14_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 425.685822609 |
| GSM99293 | H15_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 408.2530014686667 |
| GSM99294 | H15_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 315.2906994623333 |
| GSM99295 | H16_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 271.879665483 |
| GSM99296 | H16_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 373.9577281089999 |
| GSM99298 | H17_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 626.9708825309999 |
| GSM99299 | H17_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 566.4483974646666 |
| GSM99301 | H18_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 426.62092702399997 |
| GSM99302 | H18_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 444.5646583916667 |
| GSM99303 | H19_P_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 385.8206251543333 |
| GSM99304 | H19_P_B2 | male CABG patient, atrial tissue, propofol anesthesia, after CABG surgery | 512.0840146646666 |
| GSM99305 | H20_S_B1 | male CABG patient, atrial tissue, baseline before CABG surgery | 526.1352032326666 |
| GSM99306 | H20_S_B2 | male CABG patient, atrial tissue, sevoflurane anesthesia, after CABG surgery | 483.32611847766674 |
| ID | GSE56256 |
| Title | Influence of hexafluoro-2-propanol (HFIP) on lipopolysaccharides-induced inflammation in human microvascular endothelial cells of the lung |
| Organism | Homo sapiens |
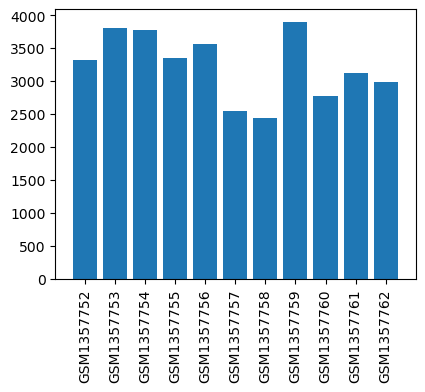
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM1357752 | 7_HFIP | cell type: Human microvascular endothelial cells (HMVEC) exposed to: HFIP 8mmol/L over a time period of 6 hrs | 3326.5375000000004 |
| GSM1357753 | 8_HFIP | cell type: Human microvascular endothelial cells (HMVEC) exposed to: HFIP 8mmol/L over a time period of 6 hrs | 3801.7225 |
| GSM1357754 | 9_HFIP | cell type: Human microvascular endothelial cells (HMVEC) exposed to: HFIP 8mmol/L over a time period of 6 hrs | 3771.5150000000003 |
| GSM1357755 | 4_LPS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: LPS 20mcg/mL over a time period of 6 hrs | 3349.907 |
| GSM1357756 | 5_LPS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: LPS 20mcg/mL over a time period of 6 hrs | 3565.1715000000004 |
| GSM1357757 | 6_LPS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: LPS 20mcg/mL over a time period of 6 hrs | 2548.363 |
| GSM1357758 | 11_LPS-HFIP | cell type: Human microvascular endothelial cells (HMVEC) exposed to: LPS 20mcg/mL and HFIP 8mmol/L over a time period of 6 hrs | 2442.562 |
| GSM1357759 | 12_LPS-HFIP | cell type: Human microvascular endothelial cells (HMVEC) exposed to: LPS 20mcg/mL and HFIP 8mmol/L over a time period of 6 hrs | 3897.2035 |
| GSM1357760 | 1_PBS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: PBS (control) over a time period of 6 hrs | 2776.605 |
| GSM1357761 | 2_PBS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: PBS (control) over a time period of 6 hrs | 3117.721 |
| GSM1357762 | 3_PBS | cell type: Human microvascular endothelial cells (HMVEC) exposed to: PBS (control) over a time period of 6 hrs | 2987.6375 |
| ID | GSE59526 |
| Title | Affymetrix Chip data of the transcriptome of human clinical study for acupuncture mechanisms on rheumatoid arthritis |
| Organism | Homo sapiens |
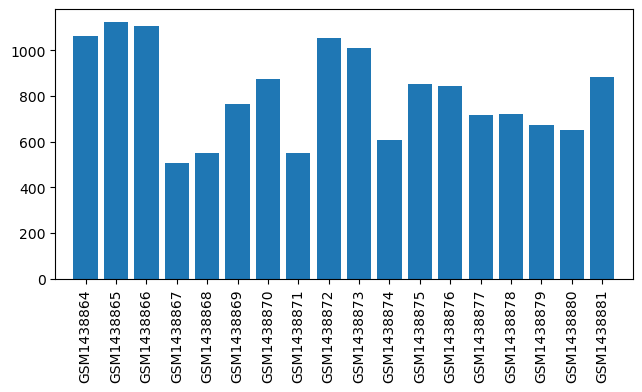
| GSM | Sample info | Characteristics | Value |
|---|---|---|---|
| GSM1438864 | Peripheral blood mononuclear cells from AP group, Patient 2 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 53 treatment group: acupuncture+methotrexate time point: before therapy patient id: 2 | 1062.6918921476665 |
| GSM1438865 | Peripheral blood mononuclear cells from AP group, Patient 3 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 48 treatment group: acupuncture+methotrexate time point: before therapy patient id: 3 | 1124.8822076666668 |
| GSM1438866 | Peripheral blood mononuclear cells from AP group, Patient 6 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 36 treatment group: acupuncture+methotrexate time point: before therapy patient id: 6 | 1105.7825691133332 |
| GSM1438867 | Peripheral blood mononuclear cells from AP group, Patient 8 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 60 treatment group: acupuncture+methotrexate time point: before therapy patient id: 8 | 506.8706561066667 |
| GSM1438868 | Peripheral blood mononuclear cells from AP group, Patient 9 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 31 treatment group: acupuncture+methotrexate time point: before therapy patient id: 9 | 548.9795034766667 |
| GSM1438869 | Peripheral blood mononuclear cells from MTX group, Patient 4 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 61 treatment group: methotrexate time point: before therapy patient id: 4 | 765.0138689233335 |
| GSM1438870 | Peripheral blood mononuclear cells from MTX group, Patient 5 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 49 treatment group: methotrexate time point: before therapy patient id: 5 | 876.8492941733334 |
| GSM1438871 | Peripheral blood mononuclear cells from MTX group, Patient 7 before therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 35 treatment group: methotrexate time point: before therapy patient id: 7 | 549.5990630833334 |
| GSM1438872 | Peripheral blood mononuclear cells from Patient 1 after 2 weeks acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 64 treatment group: acupuncture+methotrexate time point: after 2 weeks therapy patient id: 1 | 1056.1583921553333 |
| GSM1438873 | Peripheral blood mononuclear cells from Patient 3 after 2 weeks acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 48 treatment group: acupuncture+methotrexate time point: after 2 weeks therapy patient id: 3 | 1011.3653911666667 |
| GSM1438874 | Peripheral blood mononuclear cells from Patient 1 after 3 months acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 64 treatment group: acupuncture+methotrexate time point: after 3 months therapy patient id: 1 | 607.6739767466667 |
| GSM1438875 | Peripheral blood mononuclear cells from Patient 2 after 3 months acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 53 treatment group: acupuncture+methotrexate time point: after 3 months therapy patient id: 2 | 851.4977489183334 |
| GSM1438876 | Peripheral blood mononuclear cells from Patient 6 after 3 months acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 36 treatment group: acupuncture+methotrexate time point: after 3 months therapy patient id: 6 | 846.4681683643333 |
| GSM1438877 | Peripheral blood mononuclear cells from Patient 8 after 3 months acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 60 treatment group: acupuncture+methotrexate time point: after 3 months therapy patient id: | 715.38673337 |
| GSM1438878 | Peripheral blood mononuclear cells from Patient 9 after 3 months acupuncture therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 31 treatment group: acupuncture+methotrexate time point: after 3 months therapy patient id: 9 | 722.6281734366667 |
| GSM1438879 | Peripheral blood mononuclear cells from Patient 4 after 3 months MTX therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 61 treatment group: methotrexate time point: after 3 months therapy patient id: 4 | 672.48086581 |
| GSM1438880 | Peripheral blood mononuclear cells from Patient 5 after 3 months MTX therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 49 treatment group: methotrexate time point: after 3 months therapy patient id: 5 | 650.0479387766667 |
| GSM1438881 | Peripheral blood mononuclear cells from Patient 7 after 3 months MTX therapy | cell type: peripheral blood mononuclear cells disease state: rheumatoid arthritis gender: female age: 35 treatment group: methotrexate time point: after 3 months therapy patient id: 7 | 883.3097118810001 |

